This is a project the Australian community should be incredibly proud of, designed for individuals diagnosed with Angelman syndrome and those working towards research and treatments.
There are over 2400 parti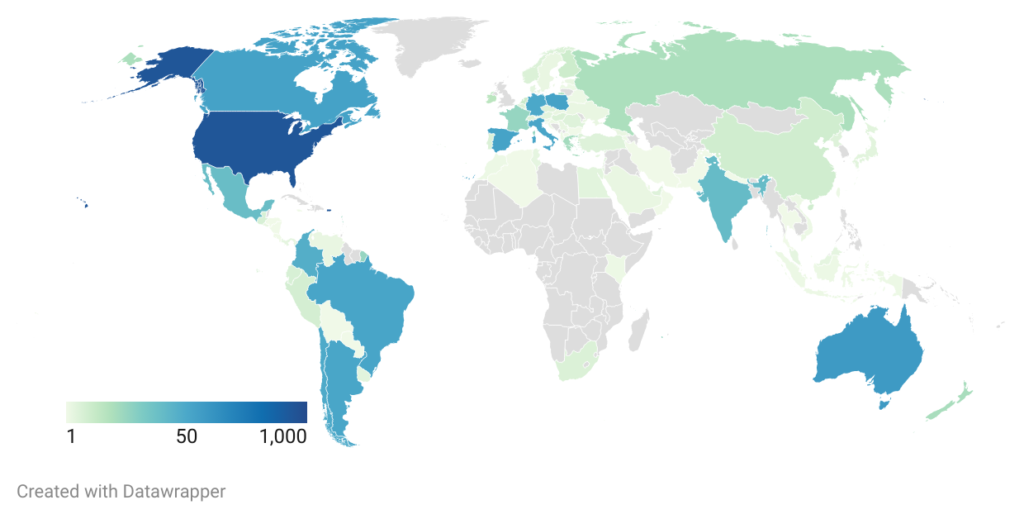 cipants across 94 different countries. We have been involved in eight different publications and the Registry is available, fully translated in seven different languages with an additional 14 partially translated. From 2016, donor funds have supported this project; development, hosting and Megan Tones’ position as data curator. This year we have received a grant from FAST United States to enable further development and support ongoing costs.
cipants across 94 different countries. We have been involved in eight different publications and the Registry is available, fully translated in seven different languages with an additional 14 partially translated. From 2016, donor funds have supported this project; development, hosting and Megan Tones’ position as data curator. This year we have received a grant from FAST United States to enable further development and support ongoing costs.
We are Growing Stronger: We’re delighted to announce the addition of a new data curator, Niki Armstrong to our team, thanks to support from FAST US. Niki will join the team soon and work alongside Megan Tones who has been doing excellent work with the registry to date. Niki brings her expertise in working with the Patient Project Muscular Dystrophy project as the Registry curator. Stay tuned for an official announcement on our socials when Niki is officially onboard.
Collaborations: You may be aware of the FAST project, spearheaded by FAST in the United States and rolled out to the global chapters and ambassadors around the world; Search & Rescue, what you may not know is that this project utilises the Registry framework, it operates under the existing ethics and governance and provides data to patient organisations around the world to ensure industry partners and patient organisations have an understanding of where the global population is. Critical Path Institute (C-Path) is another collaboration using Registry data, Our data is now live on an FDA-endorsed platform, enhancing our exposure and contributing to the creation of better outcome measures for trials. Critical Path Institute’s Rare Disease Cures Accelerator-Data and Analytics Platform (RDCA-DAP®) is an FDA-funded initiative that provides a centralized and standardized infrastructure to support and accelerate rare disease characterization, to accelerate the development of treatments and cures for rare diseases. Click here to learn more.
A New Clinic Module: Our efforts to build a clinic module are in full swing. This module will enable us to gather clinic data to complement the rich caregiver-entered dataset and allow future clinic visits to be streamlined by utilisation of existing data (you know we all hate being asked the same questions over and over again)
Utilising AI: the capabilities of AI are growing every day. The capacity to use the data we collect to create trajectories to understand how AS changes over time with different genotypes and based on medications and therapies listed would be an incredibly valuable resource that may ultimately empower our children to become their own controls in trials, potentially eliminating the need for placebos.
If you are already a participant in this project, thank you! Be sure to make sure you check your dashboard for updates that you can complete (it’s important to understand how your loved one progresses over time to build whats often referred to as “natural history” or “longitudinal data”, this is really where the magic happens, it’s not just a point in time its a record of symptoms, development and management. If you are not already in this project, please visit the website (www.angelmanregistry.info) for more information or reach out to the curator if you have any questions; curator@angelmanregistry.info.